The Vexing Problem of Wavefunction Collapse
"I think I can safely say that nobody understands quantum mechanics." ~Feynman
There are many serious and vexing problems in life. In quantum mechanics (QM), one of the most vexing is the question of wavefunction collapse. It’s also called the question of measurement. Much of what makes it vexing is that it’s behind there are so many interpretations of QM.
Interpretations are all we have because “no one understands quantum mechanics.”
Including me, so absolutely take this post with a shaker of salt. That said, most of what I’ll say you’ll find in almost any QM textbook. I’ll label my wild ass guesses explicitly.
What Is a Wavefunction?
Talking about wavefunction collapse requires an understanding of a wavefunction. The bad news is that they’re a big part of what no one understands. We don’t know the ontology — the physical reality (if any) — of the wavefunction. What we can say is that, at the very least:
A wavefunction is a mathematical function that models every aspect of a given quantum system. For any point in space, at any time, the wavefunction gives the probability of a measurement outcome at that point in space and time.
As a simple example, the wavefunction for the single electron of a hydrogen atom, among other things, gives us the probability of finding the electron at a point in space and time (where and when we look). Or it could give us the probability of the electron having a given momentum, should we measure that instead. The wavefunction describes the entire system and embodies both position and momentum information.1
Every quantum system has a wavefunction. Working with such systems requires first figuring out a mathematical wavefunction that correctly describes the particular quantum system.
Note that these wavefunctions are for quantum systems, not classical systems. Quantum mechanics and classical mechanics use different, albeit recognizably similar, mathematics. For instance, students familiar with the math of classical rotation see similar math for quantum spin.
Another big question in QM is whether classical systems are actually quantum systems with quantum wavefunctions. Many in the field believe that even classical objects, including the entire universe, have wavefunctions and are, despite their classical behavior, still quantum objects.
In a sense, this is necessarily true.2 The classical world is comprised strictly of quantum objects. As a common phrase goes, “everything is quantum.” I think the question is whether the quantum behavior of classical objects makes sense or is completely swamped out by the huge numbers of quantum systems participating.
Imagine the largest fullest stadium you’ve ever experienced. Now imagine a million of them mashed into one megastructure. Billions of people are singing the same song in unison (as best as billions can — giant electronic displays help keep them in sync). Now try to imagine picking out a 1000-person choir amongst them (let alone a single person). The whole swamps out the parts and becomes something different.
A mere gram3 of iron has roughly 10²² atoms. That’s a one followed by 22 zeros (10 sextillion). Something the weight of a paperclip has one thousand billion-billion atoms. Imagine how many voices are singing in much larger objects (like us).
So, while a large (macro) object no doubt has a wavefunction in theory, I believe that putative wavefunction contains too much information, too many voices, to be in any way meaningful. A single mathematical function representing so much information doesn’t seem possible. Information overload aside, that many data points are self-leveling or self-averaging.
Further, as mentioned, we don’t know what, if any, physical reality the wavefunction has, so there is a question of where all the information is. Presumably, in the physical world somewhere, but how does something reducible to a mathematical function describe, say, a chair (let alone all of reality).
What Is Wavefunction Collapse?
A wavefunction gives the probability of measuring some quantum property. For instance, an electron’s location in space and time. In fact, a wavefunction gives the probability of finding the electron at all points and times.
And until we actually look for it — and either find it there or don’t — the particle is in a superposition of probabilities for being in all possible places (and times).
Experiments seem to demonstrate that, contrary to what is often said, it’s not so much the particle is “everywhere at once” but that, in some real sense, it has no location until we localize it (by finding it).
And therein lies the rub. When we do find the particle — when we localize it — its wavefunction “collapses” (or “reduces”) from the superposition of ‘anywhere’ to ‘right here’. This is a problem. A big one. The problem of wavefunction collapse.
The smaller issue is that the equations we use with wavefunctions (the Schrödinger equation, the Kline-Gordon equation, and the Dirac equation) don’t account for this sudden change from diffuse to specific. There is no math (so far!) telling us how to reduce the wavefunction from a superposition of many states to known single state. The problem goes beyond some missing math to what Einstein really meant by “spooky action at a distance.”
Here’s the Big Vexing Problem of wavefunction collapse: The wavefunction specifies a particle’s position probability for every point in the universe. Throughout most of the universe the probability is effectively zero. But not zero exactly. So, the wavefunction is telling us that there is some chance the particle could be literally anywhere.
But when the wavefunction “collapses” then the probability spread throughout the universe instantly vanishes. We know where the particle is (we just found it), so for the moment there is exactly zero probability of finding it anywhere else. It’s the “instantly” that’s the problem. Information isn’t allowed to be infinitely fast, not in this universe.
But (as always) It Depends
If the wavefunction is only a mathematical function with no physical representation — if it just represents our knowledge of the system — then this spooky instant vanishing act isn’t a big deal. We thought the particle might be way over there, but now we know it isn’t. All that’s changed is our knowledge.
But if the wavefunction corresponds with some physical aspect of the real world, and there are good reasons to think it does, then this instant change is a huge problem. Einstein’s Spook. Along with associated effects, it is, in some regards, the central unsolved problem of quantum mechanics.
It’s what drives the infamous Many Worlds Interpretation (the MWI) of quantum mechanics, which denies that collapse ever happens. Rather than collapsing, the wavefunction continues but branches to describe both outcomes.4
Some interpretations of QM largely ignore the issue because quantum mechanics works. Who cares if we don’t understand why. Use it to do cool stuff and let someone else worry about the philosophy. Most of the world probably falls into this category.
But others seek to explain collapse, to find that missing math. One direction for this is objective-collapse theories. One I think sounds promising is the Diósi–Penrose model, which factors in gravity — which is currently excluded from QM entirely.5
My Wild Ass Guess
I believe in what’s called the Heisenberg Cut — the hypothetical division between the quantum and classical worlds. I think quantum behavior is swamped out and constantly collapsing (and I like the idea that gravity might be involved).
I speculate there is a connection between an object’s de Broglie wavelength and the ontology of a wavefunction. Quantum objects are wavelets in quantum fields, so we’re in a decidedly wave-based regime.6 There is a fundamental unanswered question about how an electron is able to interfere with itself in single-electron double-slit experiments. What exactly goes through both slits and interacts with itself? Mathematically, the wavefunction, but how does reality do it?
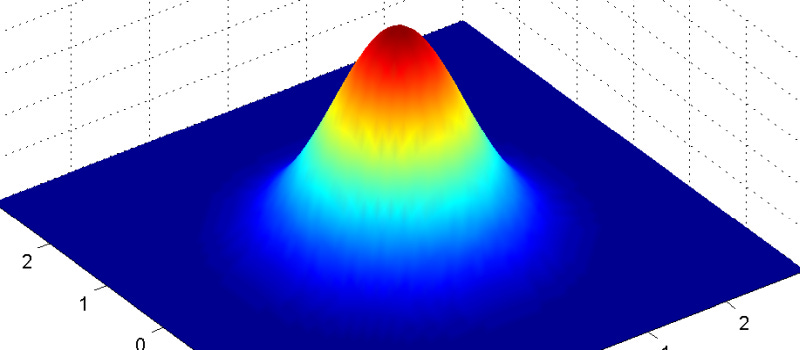
We say the electron “could be anywhere” (which is literally true), but the actuality is that it’s most likely somewhere near where we expect. Especially if we just localized it by finding it. But the longer we leave the electron alone (unobserved), the more its wavefunction spreads out. This makes us increasingly less certain of its location as time progresses. The bump in the image above flattens out over time.
But the energy/mass of the electron is a single quantum of the electron field, so what does it mean for it to be spread out? How does the indivisible unit of energy/mass get distributed? How, or what, passes through the two slits? More critically, how does that distribution “collapse” instantly into a point? (In apparent defiance of our understanding of the universal speed limit.)
To me that seems the central mystery to solve, and I WAG it might be connected with the quantum nonlocality we see in Bell’s Test experiments with entangled particles. These experiments demonstrate that the quantum world does operate in a nonlocal fashion that doesn’t violate the speed law (because it can’t be used to send information). Perhaps this same quantum nonlocality pertains to the distributed energy/mass instantly relocating to one location. Perhaps because the energy/mass is sub-quantum for the field the usual rules don’t apply.
I envision the wavelets representing quantum particles to — exactly as our equations suggest — spread out continually. When some part of that spread out energy wavelet finds reason to interact with another quantum wavelet, the point-like interaction instantly “drains” the spread-out energy into the other particle or into new particles the interaction creates.
For instance, suppose an electron encounters an anti-electron (a positron). This results in the annihilation of both and the creation of two high-energy photons (gamma ray “particles”). All the energy of the first two goes into creating the second two.
Alternately, a photon from a laser cat teaser might be absorbed by an electron in the rug or wall. In this case, all the energy of the photon is drained into the electron, which boosts it into a higher energy orbit.
Either way, the mystery is how the supposedly spread-out energy instantly changes, and I think this may be related to quantum nonlocality. We may need to accept that the quantum world isn’t restricted to the same speed limits as the classical world.
Which may itself be another instance of the Heisenberg Cut.
But, as I said, take all this with a large shaker of salt.
Until next time…
More realistic wavefunctions must include other measurable quantum properties, such as quantum spin.
In my opinion, only in a sense, but not really.
Canonically, the weight of an ordinary paperclip.
I may write a post about all the objections I have to the MWI, especially if there’s an interest, but for now suffice to say I’m not sympathetic to any multiverse theory.
Which is another Major Problem in quantum physics. How to we unify our two greatest theories, QM and General Relativity? They don’t get along at all.
In the current view, known as Quantum Field Theory (QFT), there are no particles, just point-like interactions. When we localize an electron, it’s the interaction that is local.